Paper:
Deformation and Trapping of Cell Nucleus Using Micropillar Substrates Possibly Affect UV Radiation Resistance of DNA
Kazuaki Nagayama, Chiaki Sagawa, and Akiko Sato
Micro-Nano Biomechanics Laboratory, Department of Mechanical Systems Engineering, Ibaraki University
4-12-1 Nakanarusawa-cho, Hitachi, Ibaraki 316-8511, Japan
DNA damage induced by the ultraviolet (UV) light, which affects adversely on genome stability, causes many kinds of diseases. Thus, a biochemical or biomechanical method in DNA damage protection is well required. In the present study, we investigated the effects of mechanical factors, such as deformation of cell nucleus using polydimethylsiloxane (PDMS)-based microfabricated array of micropillars, on UV radiation resistance of DNA in cultured cells. The epithelial-like cells spread normally in the spaces between micropillars and their nuclei showed remarkable deformation and appeared to be “trapped” mechanically on the array of pillars. We found that the UV radiation-induced DNA damage estimated by the fluorescent intensity of the phospho-histone γ-H2AX, was significantly inhibited with the nucleus deformation on the pillars. The result indicates that the inhibition of UV radiation-induced DNA damages might be resulted from structural change of DNA caused by the mechanical stress of the cell nucleus on the micropillars.
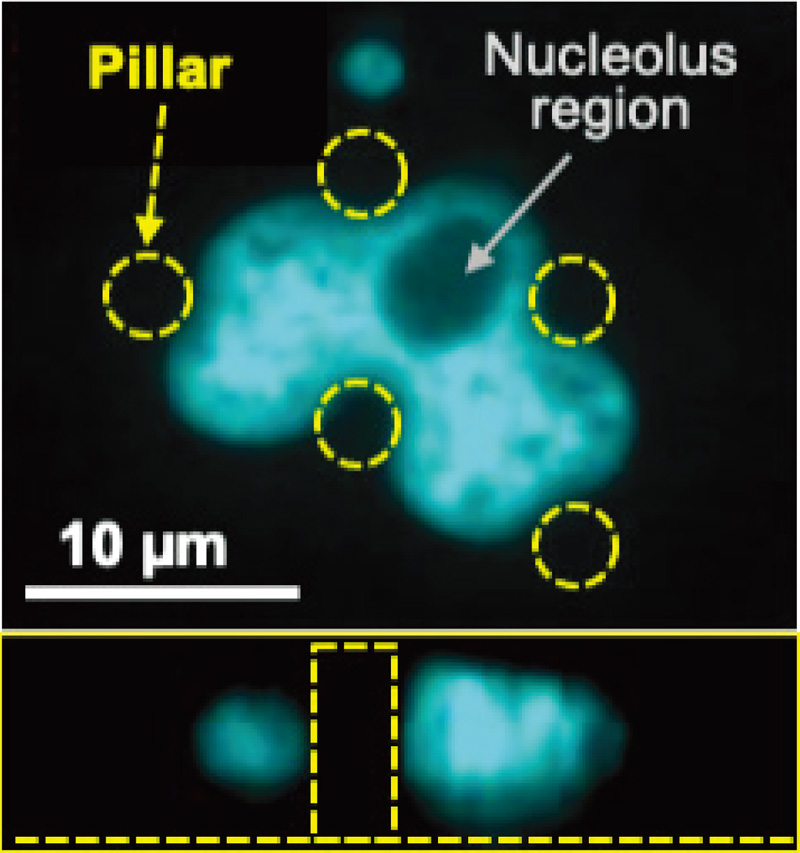
Cell nucleus deformation on micropillars
- [1] T. G. Gorgels and D. van Norren, “Ultraviolet and green light cause different types of damage in rat retina,” Invest. Ophthalmol. Vis. Sci., Vol.36, No.5, pp. 851-863, 1995.
- [2] M. Rózanowska, J. Jarvis-Evans, W. Korytowski, M. E. Boulton, J. M. Burke, and T. Sarna, “Blue light induced reactivity of retinal age pigment: In vitro generation of oxygen-reactive species,” J. Biol. Chem., Vol.270, No.32, pp. 18825-18830, 1995. https://doi.org/10.1074/jbc.270.32.18825
- [3] A. Stary, C. Robert, and A. Sarasin, “Deleterious effects of ultraviolet A radiation in human cells,” Mutat. Res., Vol.383, No.1, pp. 1-8, 1997. https://doi.org/10.1016/s0921-8777(96)00041-9
- [4] C. Reme, J. Reinboth, M. Clausen, and F. Hafezi, “Light damage revisited: Converging evidence, diverging views?,” Graefes. Arch. Clin. Exp. Ophthalmol., Vol.234, pp. 2-11, 1996.
- [5] C. Alapetite, T. Wachter, E. Sage, and F. Moustachi, “The use of the comet assay to detect DNA-repair deficiencies in human fibroblasts exposed to UVC, UVB, UVA and gamma-rays,” Int. J. Radiat. Biol., Vol.69, No.3, pp. 359-369, 1996. https://doi.org/10.1080/095530096145922
- [6] C. Keilbassa, L. Roza, and B. Epe, “Wavelength dependence of oxidative DNA damage induced by UV and visible light,” Carcinogenesis, Vol.18, No.4, pp. 811-816, 1997. https://doi.org/10.1093/carcin/18.4.811
- [7] D. E. Ingber, “Fibronectin controls capillary endothelial cell growth by modulating cell shape,” Proc. Natl. Acad. Sci. USA, Vol.87, No.9, pp. 3579-3583, 1990. https://doi.org/10.1073/pnas.87.9.3579
- [8] A. J. Engler, S. Sen, H. L. Sweeney, and D. E. Discher, “Matrix elasticity directs stem cell lineage specification,” Cell, Vol.126, No.4, pp. 677-689, 2006. https://doi.org/10.1016/j.cell.2006.06.044
- [9] C. S. Chen, M. Mrksich, S. Huang, G. M. Whitesides, and D. E. Ingber, “Geometric control of cell life and death,” Science, Vol.276, No.5317, pp. 1425-1428, 1997. https://doi.org/10.1126/science.276.5317.1425
- [10] P. G. Smith, C. Roy, S. Fisher, Q. Q. Huang, and F. Brozovich, “Selected contribution: Mechanical strain increases force production and calcium sensitivity in cultured airway smooth muscle cells,” J. Appl. Physiol., Vol.89, No.5, pp. 2092-2098, 2000. https://doi.org/10.1152/jappl.2000.89.5.2092
- [11] K. N. Dahl, A. J. S. Ribeiro, and J. Lammerding, “Nuclear shape, mechanics, and mechanotransduction,” Circ. Res., Vol.102, No.11, pp. 1307-1318, 2008. https://doi.org/10.1161/CIRCRESAHA.108.173989
- [12] M. L. Lombardi, D. E. Jaalouk, C. M. Shanahan, B. Burke, K. J. Roux, and J. Lammerding, “The interaction between nesprins and sun proteins at the nuclear envelope is critical for force transmission between the nucleus and cytoskeleton,” J. Biol. Chem., Vol.286, No.30, pp. 26743-26753, 2011. https://doi.org/10.1074/jbc.M111.233700
- [13] P. Roca-Cusachs, J. Alcaraz, R. Sunyer, J. Samitier, R. Farré, and D. Navajas, “Micropatterning of single endothelial cell shape reveals a tight coupling between nuclear volume in G1 and proliferation,” Biophys. J., Vol.94, No.12, pp. 4984-4995, 2008. https://doi.org/10.1529/biophysj.107.116863
- [14] M. Versaevel, T. Grevesse, and S. Gabriele, “Spatial coordination between cell and nuclear shape within micropatterned endothelial cells,” Nat. Commun., Vol.3, Article No.671, 2012. https://doi.org/10.1038/ncomms1668
- [15] K. Nagayama, Y. Hamaji, Y. Sato, and T. Matsumoto, “Mechanical trapping of the nucleus on micropillared surfaces inhibits the proliferation of vascular smooth muscle cells but not cervical cancer HeLa cells,” J. Biomech., Vol.48, No.10, pp. 1796-1803, 2015. https://doi.org/10.1016/j.jbiomech.2015.05.004
- [16] K. Nagayama, “Mechanical stress to cell nucleus inhibits proliferation and differentiation of vascular smooth muscle cells,” J. Biosci. Medic., Vol.8, No.5, pp. 132-141, 2020. https://doi.org/10.4236/jbm.2020.85013
- [17] J. L. Tan, J. Tien, D. M. Pirone, D. S. Gray, K. Bhadriraju, and C. S. Chen, “Cells lying on a bed of microneedles: an approach to isolate mechanical force,” Proc. Natl. Acad. Sci. USA, Vol.100, No.4, pp. 1484-1489, 2003. https://doi.org/10.1073/pnas.0235407100
- [18] N. Sakamoto, Y. Yamazaki, T. Ohashi, and M. Sato, “Effect of Mechanical Environment of Focal Adhesions on Remodeling of Endothelial Cells Subjected to Cyclic Stretching Using Microsubstrates,” J. Robot. Mechatron., Vol.19, No.5, pp. 557-564, 2007. https://doi.org/10.20965/jrm.2007.p0557
- [19] K. Nagayama and T. Matsumoto, “Dynamic change in morphology and traction forces at focal adhesions in cultured vascular smooth muscle cells during contraction,” Cell. Mol. Bioeng., Vol.4, pp. 348-357, 2011. https://doi.org/10.1007/s12195-011-0166-y
- [20] B. Zeng, S. Tong, X. Ren, and H. Xia, “Cardiac cell proliferation assessed by EdU, a novel analysis of cardiac regeneration,” Cytotechnology, Vol.68, No.4, pp. 763-770, 2016. https://doi.org/10.1007/s10616-014-9827-8
- [21] R. E. Krisch, M. B. Flick, and C. N. Trumbore, “Radiation chemical mechanisms of single- and double-strand break formation in irradiated SV40 DNA,” Radiat. Res., Vol.126, No.2, pp. 251-259, 1991. https://doi.org/10.2307/3577826
- [22] H. Takata, T. Hanafusa, T. Mori, M. Shimura, Y. Iida, K. Ishikawa, K. Yoshikawa, Y. Yoshikawa, and K. Maeshima, “Chromatin compaction protects genomic DNA from radiation damage,” PLoS One, Vol.8, No.10, Article No.e75622, 2013. https://doi.org/10.1371/journal.pone.0075622
- [23] Y. Yoshikawa, T. Mori, N. Magome, K. Hibino, and K. Yoshikawa, “DNA compaction plays a key role in radioprotection against double-strand breaks as revealed by single-molecule observation,” Chem. Phys. Lett., Vol.456, Issues 1-3, pp. 80-83, 2008. https://doi.org/10.1016/j.cplett.2008.03.009
 This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.
This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.