Review:
Impact of the Upper Limb Physiotherapy on Behavioral and Brain Adaptations in Post-Stroke Patients
Wataru Kuwahara*,**, Yu Miyawaki**,***, and Fuminari Kaneko*,**
*Department of Rehabilitation Medicine, Keio University School of Medicine
35 Shinanomachi, Shinjuku-ku, Tokyo 160-8582, Japan
**Department of Physical Therapy, Graduate School of Health Sciences, Tokyo Metropolitan University
7-2-10 Higashi-Oku, Arakawa-ku, Tokyo 116-8551, Japan
***Human Augmentation Research Center, National Institute of Advanced Industrial Science and Technology
6-2-3 Kashiwanoha, Kashiwa, Chiba 277-0882, Japan
Many stroke patients suffer from motor impairments due to paralysis, and consequently, motor paralysis of upper limbs seems to be particularly prone to residual impairment compared to that of lower limbs. Although ‘learned non-use’ that by managing reasonably well using only the unaffected upper limb in their actions, the patients can achieve their desired behavior, and these success experiences strengthen this pattern of behavior can be interpreted as a post-stroke adaptation, physiotherapy may lead to poor recovery of motor impairment. This review article discusses the impact of upper limb physiotherapy after stroke on behavioral/brain adaptations. Our previous studies demonstrated that patients with severe post-stroke sensorimotor impairments in a chronic phase might have abnormal functional connectivity. To prevent such adaptation after stroke, upper limb physiotherapy is important. In rehabilitation practices, hyper-adaptation has been often observed in not only behavioral but also brain changes. Although several studies are reporting clinical efficacy in patients with moderate to mild paralysis, there might be no effective treatment for patients with severe motor paralysis. To overcome these serious problems, we have developed a novel approach, kinesthetic illusion induced by visual stimulation (KINVIS) therapy. We showed that the effects of KINVIS therapy with therapeutic exercise on upper limb motor functions were mediated by spasticity, and functional connectivity in the brain was also changed with the improvement of motor function after KINVIS therapy. Brain changes underlying behavioral changes need to be more examined, and the adaptation of stroke patients needs to be clarified in detail.
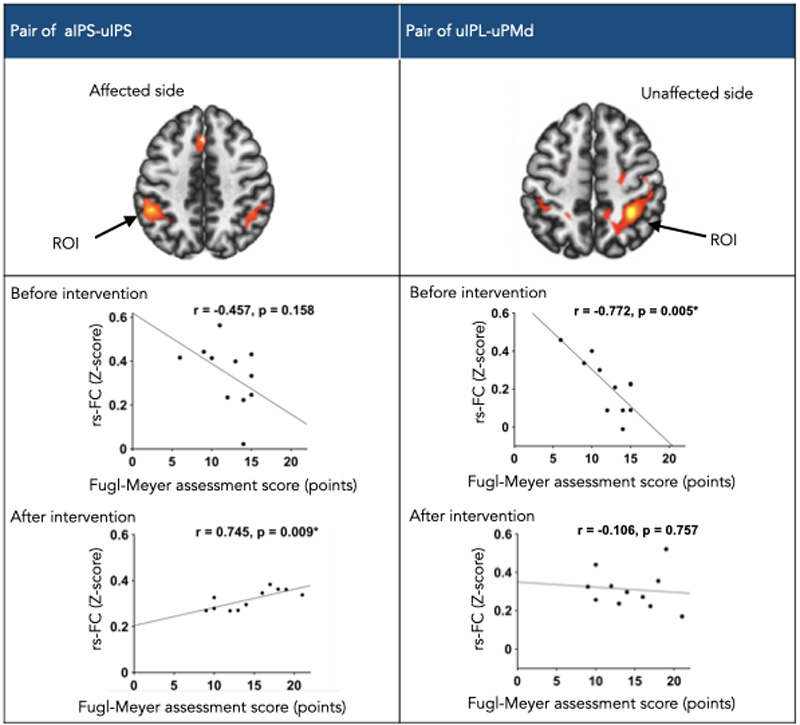
Brain adaptation in post-stroke patients
- [1] N. Takashima, H. Arima, Y. Kita, T. Fujii, N. Miyamatsu, M. Komori, Y. Sugimoto, S. Nagata, K. Miura, and K. Nozaki, “Incidence, Management and Short-Term Outcome of Stroke in a General Population of 1.4 Million Japanese – Shiga Stroke Registry –,” Circ. J., Vol.81, No.11, pp. 1636-1646, 2017.
- [2] M. P. Lindsay, B. Norrving, R. L. Sacco, M. Brainin, W. Hacke, S. Martins, J. Pandian, and V. Feigin, “World Stroke Organization (WSO): Global Stroke Fact Sheet 2019,” Int. J. of Stroke., Vol.14, No.8, pp. 806-817, 2019.
- [3] H. T. Hendricks, J. van Limbeek, A. C. Geurts, and M. J. Zwarts, “Motor recovery after stroke: a systematic review of the literature,” Arch. Phys. Med. Rehabil., Vol.83, No.11, pp. 1629-1637, 2002.
- [4] M. F. Levin, J. A. Kleim, and S. L. Wolf, “What do motor “recovery” and “compensation” mean in patients following stroke?,” Neurorehabil. Neural Repair., Vol.23, No.4, pp. 313-319, 2009.
- [5] L. M. Wiklund and P. Uvebrant, “Hemiplegic cerebral palsy: correlation between CT morphology and clinical findings,” Dev. Med. Child Neurol., Vol.33, No.6, pp. 33512-523, 1991.
- [6] E. M. A. El-Kafy, M. A. Alshehri, A. A. R. El-Fiky, and M. A. Guermazi, “The Effect of Virtual Reality-Based Therapy on Improving Upper Limb Functions in Individuals With Stroke: A Randomized Control Trial,” Front. Aging Neurosci., Vol.13, Article No.731343, 2021.
- [7] E. Taub, G. Uswatte, and T. Elbert, “New treatments in neurorehabilitation founded on basic research,” Nat. Rev. Neurosci., Vol.3, No.3, pp. 228-236, 2002.
- [8] E. Taub, G. Uswatte, and D. M. Morris, “Improved motor recovery after stroke and massive cortical reorganization following Constraint-Induced Movement therapy,” Phys. Med. Rehabil. Clin. N. Am., Vol.14, Issue 1, Supplement, pp. S77-S91, 2003.
- [9] N. Murase, J. Duque, R. Mazzocchio, and L. G. Cohen, “Influence of interhemispheric interactions on motor function in chronic stroke,” Ann. Neurol., Vol.55, No.3, pp. 400-409, 2004.
- [10] D. A. Nowak, C. Grefkes, M. Dafotakis, S. Eickhoff, J. Küst, H. Karbe, and G. R. Fink, “Effects of low-frequency repetitive transcranial magnetic stimulation of the contralesional primary motor cortex on movement kinematics and neural activity in subcortical stroke,” Arch. Neurol., Vol.65, No.6, pp. 741-747, 2008.
- [11] R. J. Nudo and G. W. Milliken, “Reorganization of movement representations in primary motor cortex following focal ischemic infarcts in adult squirrel monkeys,” J. Neurophysiol., Vol.75, No.5, pp. 2144-2149, 1996.
- [12] P. W. Duncan, L. B. Goldstein, D. Matchar, G. W. Divine, and J. Feussner, “Measurement of motor recovery after stroke. Outcome assessment and sample size requirements,” Stroke, Vol.23, No.8, pp. 1084-1089, 1992.
- [13] H. Nakayama, H. S. Jørgensen, H. O. Raaschou, and T. S. Olsen, “Recovery of upper extremity function in stroke patients: the Copenhagen Stroke Study,” Arch. Phys. Med. Rehabil., Vol.75, No.4, pp. 394-398, 1994.
- [14] R. M. Dijkhuizen, J. Ren, J. B. Mandeville, O. Wu, F. M. Ozdag, M. A. Moskowitz, B. R. Rosen, and S. P. Finklestein, “Functional magnetic resonance imaging of reorganization in rat brain after stroke,” Proc. Natl. Acad. Sci. U.S.A., Vol.98, No.22, pp. 12766-12771, 2001.
- [15] R. Weber, P. Ramos-Cabrer, C. Justicia, D. Wiedermann, C. Strecker, C. Sprenger, and M. Hoehn, “Early prediction of functional recovery after experimental stroke: functional magnetic resonance imaging, electrophysiology, and behavioral testing in rats,” J. Neurosci., Vol.28, No.5, pp. 1022-1029, 2008.
- [16] R. M. Dijkhuizen and K. Nicolay, “Magnetic resonance imaging in experimental models of brain disorders,” J. Cereb. Blood Flow Metab., Vol.23, pp. 1383-1402, 2003.
- [17] I. Loubinoux, C. Carel, J. Pariente, S. Dechaumont, J.-F. Albucher, P. Marque, C. Manelfe, and F. Chollet, “Correlation between cerebral reorganization and motor recovery after subcortical infarcts,” Neuroimage, Vol.20, No.4, pp. 2166-2180, 2003.
- [18] D. Tombari, I. Loubinoux, J. Pariente, A. Gerdelat, J.-F. Albucher, J. Tardy, E. Cassol, and F. Chollet, “A longitudinal fMRI study: in recovering and then in clinically stable sub-cortical stroke patients,” Neuroimage, Vol.23, No.3, pp. 827-839, 2004.
- [19] N. S. Ward, M. M. Brown, A. J. Thompson, and R. S. J. Frackowiak, “Neural correlates of outcome after stroke: a cross-sectional fMRI study,” Brain, Vol.126, Part 6, pp. 1430-1448, 2003.
- [20] T. G. Hornby, D. S. Reisman, I. G. Ward, P. L. Scheets, A. Miller, D. Haddad, E. J. Fox, N. E. Fritz, K. Hawkins, C. E. Henderson, K. L. Hendron, C. L. Holleran, J. E. Lynskey, and A. Walter, “Clinical Practice Guideline to Improve Locomotor Function Following Chronic Stroke, Incomplete Spinal Cord Injury, and Brain Injury,” J. Neurol. Phys. Ther, Vol.44, No.1, pp. 49-100, 2020.
- [21] R. Teasell, N. Hussein, M. Mirkowski, D. V. Rrt, M. Saikaley Hbsc, M. Longval Bsc, and J. Iruthayarajah, “Stroke Rehabilitation Clinician Handbook: 4. Hemiplegic Upper Extremity Rehabilitation,” EBRSR, 2020.
- [22] A. K. Rehme, S. B. Eickhoff, L. E. Wang, G. R. Fink, and C. Grefkes, “Dynamic causal modeling of cortical activity from the acute to the chronic stage after stroke,” Neuroimage, Vol.55, pp. 1147-1158, 2011.
- [23] Y. Miyawaki, M. Yoneta, M. Okawada, M. Kawakami, M. Liu, and F. Kaneko, “Model-Based Analyses for the Causal Relationship Between Post-stroke Impairments and Functional Brain Connectivity Regarding the Effects of Kinesthetic Illusion Therapy Combined With Conventional Exercise,” Front. Syst. Neurosci., Vol.15, Article No.804263, 2022.
- [24] T. Hanakawa, I. Immisch, K. Toma, M. A. Dimyan, P. Van Gelderen, and M. Hallett, “Functional properties of brain areas associated with motor execution and imagery,” J. Neurophysiol., Vol.89, No.2, pp. 989-1002, 2003.
- [25] T. Hanakawa, M. A. Dimyan, and M. Hallett, “Motor Planning, Imagery, and Execution in the Distributed Motor Network: A Time-Course Study with Functional MRI,” Cereb. Cortex., Vol.18, No.12, pp. 2775-2788, 2008.
- [26] M. S. A. Graziano, G. S. Yap, and C. G. Gross, “Coding of Visual Space by Premotor Neurons,” Science, Vol.266, pp. 1054-1057, 1994.
- [27] G. Rizzolatti, G. Luppino, and M. Matelli, “The organization of the cortical motor system: new concepts, Electroencephalogr,” Clin. Neurophysiol., Vol.106, pp. 283-296, 1998.
- [28] B. Kably and T. Drew, “Corticoreticular pathways in the cat. I. Projection patterns and collaterization,” J. Neurophysiol., Vol.80, No.1, pp. 389-405, 1998.
- [29] K. Matsuyama and T. Drew, “Organization of the projections from the pericruciate cortex to the pontomedullary brainstem of the cat: a study using the anterograde tracer Phaseolus vulgaris-leucoagglutinin,” J. Comp. Neurol., Vol.389, No.4, pp. 617-641, 1997.
- [30] S. S. Yeo, M. C. Chang, Y. H. Kwon, Y. J. Jung, and S. H. Jang, “Corticoreticular pathway in the human brain: Diffusion tensor tractography study,” Neurosci. Lett., Vol.508, No.1, pp. 9-12, 2012.
- [31] H. Eberle, Y. Hayashi, R. Kurazume, T. Takei, and Q. An, “Modeling of hyper-adaptability: from motor coordination to rehabilitation,” Adv. Robot., Vol.35, Issues 13-14, pp. 802-817, 2021.
- [32] S. L. Wolf, C. J. Winstein, J. P. Miller, E. Taub, G. Uswatte, D. Morris, C. Giuliani, K. E. Light, and D. Nichols-Larsen, “Effect of Constraint-Induced Movement Therapy on Upper Extremity Function 3 to 9 Months After Stroke: The EXCITE Randomized Clinical Trial,” J. of the American Medical Association, Vol.296, No.17, pp. 2095-2104, 2006.
- [33] A. P. Reiss, S. L. Wolf, E. A. Hammel, E. L. McLeod, and E. A. Williams, “Constraint-Induced Movement Therapy (CIMT): Current Perspectives and Future Directions,” Stroke Res. Treat., Article No.159391, 2012.
- [34] C. Dohle, J. Pùllen, A. Nakaten, J. Kúst, C. Rietz, and H. Karbe, “Mirror therapy promotes recovery from severe hemiparesis: A randomized controlled trial,” Neurorehabil. Neural Repair, Vol.23, No.3, pp. 209-217, doi: 10.1177/1545968308324786, 2009.
- [35] S. J. Page, P. Levine, and A. Leonard, “Mental practice in chronic stroke: results of a randomized, placebo-controlled trial,” Stroke, Vol.38, No.4, pp. 1293-1297, 2007.
- [36] V. Klamroth-Marganska, J. Blanco, K. Campen, A. Curt, V. Dietz, T. Ettlin, M. Felder, B. Fellinghauer, M. Guidali, A. Kollmar, A. Luft, T. Nef, C. Schuster-Amft, W. Stahel, and R. Riener, “Three-dimensional, task-specific robot therapy of the arm after stroke: a multicentre, parallel-group randomised trial,” Lancet Neurol., Vol.13, No.2, pp. 159-166, 2014.
- [37] A. Pollock, S. E. Farmer, M. C. Brady, P. Langhorne, G. E. Mead, J. Mehrholz, and F. van Wijck, “Interventions for improving upper limb function after stroke,” Cochrane Database Syst. Rev., No.11, Article No.CD010820, 2014.
- [38] A. Ramos-Murguialday, D. Broetz, M. Rea, L. Läer, Ö. Yilmaz, F. L. Brasil, G. Liberati, M. R. Curado, E. Garcia-Cossio, A. Vyziotis, W. Cho, M. Agostini, E. Soares, S. Soekadar, A. Caria, L. G. Cohen, and N. Birbaumer, “Brain–machine interface in chronic stroke rehabilitation: A controlled study,” Ann. Neurol., Vol.74, No.1, pp. 100-108, 2013.
- [39] A. Ramos-Murguialday, M. R. Curado, D. Broetz, Ö. Yilmaz, F. L. Brasil, G. Liberati, E. Garcia-Cossio, W. Cho, A. Caria, L. G. Cohen, and N. Birbaumer, “Brain–Machine Interface in Chronic Stroke: Randomized Trial Long-Term Follow-up,” Neurorehabil. Neural Repair., Vol.33, No.3, pp. 188-198, 2019.
- [40] R. J. Nudo, “Postinfarct cortical plasticity and behavioral recovery,” Stroke, Vol.38, Issue 2, Supplement, pp. 840-845, 2007.
- [41] T. A. Jones, C. J. Chu, L. A. Grande, and A. D. Gregory, “Motor Skills Training Enhances Lesion-Induced Structural Plasticity in the Motor Cortex of Adult Rats,” J. Neurosci., Vol.19, No.22, pp. 10153-10163, 1999.
- [42] J. Biernaskie and D. Corbett, “Enriched Rehabilitative Training Promotes Improved Forelimb Motor Function and Enhanced Dendritic Growth after Focal Ischemic Injury,” J. Neurosci., Vol.21, No.14, pp. 5272-5280, 2001.
- [43] N. Dancause, S. Barbay, S. B. Frost, E. J. Plautz, D. Chen, E. V. Zoubina, A. M. Stowe, and R. J. Nudo, “Extensive Cortical Rewiring after Brain Injury,” J. Neurosci., Vol.25, No.44, pp. 10167-10179, 2005.
- [44] J. Liepert, J. Greiner, V. Nedelko, and C. Dettmers, “Reduced upper limb sensation impairs mental chronometry for motor imagery after stroke: Clinical and electrophysiological findings,” Neurorehabil. Neural Repair., Vol.26, No.5, pp. 470-478, 2012.
- [45] C. Colomer and E. N. R. Llorens, “Mirror therapy in chronic stroke survivors with severely impaired upper limb function: a randomized controlled trial,” Eur. J. Phys. Rehabil. Med., Vol.52, No.3, pp. 271-278, 2016.
- [46] S. Carda, A. Biasiucci, A. Maesani, S. Ionta, J. Moncharmont, S. Clarke, M. M. Murray, and J. del R. Millán, “Electrically Assisted Movement Therapy in Chronic Stroke Patients With Severe Upper Limb Paresis: A Pilot, Single-Blind, Randomized Crossover Study,” Arch. Phys. Med. Rehabil., Vol.98, No.8, pp. 1628-1635, 2017.
- [47] F. Kaneko, K. Shindo, M. Yoneta, M. Okawada, K. Akaboshi, and M. Liu, “A Case Series Clinical Trial of a Novel Approach Using Augmented Reality That Inspires Self-body Cognition in Patients With Stroke: Effects on Motor Function and Resting-State Brain Functional Connectivity,” Front. Syst. Neurosci., Vol.13, 2019.
- [48] F. Kaneko, T. Yasojima, and T. Kizuka, “Kinesthetic illusory feeling induced by a finger movement movie effects on corticomotor excitability,” Neuroscience., Vol.149, No.4, pp. 976-984, 2007.
- [49] T. Aoyama, F. Kaneko, T. Hayami, and E. Shibata, “The effects of kinesthetic illusory sensation induced by a visual stimulus on the corticomotor excitability of the leg muscles,” Neurosci. Lett., Vol.514, No.1, pp. 106-109, 2012.
- [50] F. Kaneko, C. Blanchard, N. Lebar, B. Nazarian, A. Kavounoudias, and P. Romaiguère, “Brain Regions Associated to a Kinesthetic Illusion Evoked by Watching a Video of One’s Own Moving Hand,” PLoS One, Vol.10, No.8, Article No.e0131970, 2015.
- [51] E. Shibata and F. Kaneko, “Event-related desynchronization possibly discriminates the kinesthetic illusion induced by visual stimulation from movement observation,” Exp. Brain Res., Vol.237, pp. 3233-3240, 2019.
- [52] F. Kaneko, R. Takahashi, E. Shibata, and Y. Itaguchi, “Kinesthetic illusion induced by pairing of visual and peripheral nerve stimulation causes sustained enhancement of corticospinal tract excitability,” Society for Neuroscience San Diego, pp. 12-16, 2016.
- [53] F. Kaneko, T. Murakami, K. Onari, H. Kurumadani, and K. Kawaguchi, “Decreased cortical excitability during motor imagery after disuse of an upper limb in humans,” Clin. Neurophysiol., Vol.114, No.12, pp. 2397-2403, 2003.
- [54] T. Inada, F. Kaneko, and T. Hayami, “Effect of kinesthetic illusion induced by visual stimulation on muscular output function after short-term immobilization,” J. Electromyogr. Kinesiol., Vol.27, pp. 66-72, 2016.
- [55] M. Okawada, F. Kaneko, K. Shindo, M. Yoneta, K. Sakai, K. Okuyama, K. Akaboshi, and M. Liu, “Kinesthetic illusion induced by visual stimulation influences sensorimotor event-related desynchronization in stroke patients with severe upper-limb paralysis: A pilot study,” Restor. Neurol. Neurosci., Vol.38, No.6, pp. 455-465, 2020.
- [56] R. Takahashi, F. Kaneko, M. Koiwa, W. Ide, M. Okawada, and K. Akaboshi, “A prospective case series using visually induced kinaesthetic illusion on patients with chronic stroke,” J. Rehabil. Med., 2022 (in press).
- [57] F. Grimm, G. Naros, and A. Gharabaghi, “Closed-loop task difficulty adaptation during virtual reality reach-to-grasp training assisted with an exoskeleton for stroke rehabilitation,” Front. Neurosci., Vol.10, No.518, 2016.
- [58] S. C. Barros Galvão, R. Borba Costa Dos Santos, P. Borba Dos Santos, M. E. Cabral, and K. Monte-Silva, “Efficacy of Coupling Repetitive Transcranial Magnetic Stimulation and Physical Therapy to Reduce Upper-Limb Spasticity in Patients With Stroke: A Randomized Controlled Trial,” Arch. Phys. Med. Rehabil., Vol.95, No.2, pp. 222-229, 2014.
- [59] J. J. J. Chen, Y. N. Wu, S. C. Huang, H. M. Lee, and Y. L. Wang, “The Use of a Portable Muscle Tone Measurement Device to Measure the Effects of Botulinum Toxin Type A on Elbow Flexor Spasticity,” Arch. Phys. Med. Rehabil., Vol.86, No.8, pp. 1655-1660, 2005.
- [60] R. van der Vliet, R. W. Selles, E. R. Andrinopoulou, R. Nijland, G. M. Ribbers, M. A. Frens, C. Meskers, and G. Kwakkel, “Predicting Upper Limb Motor Impairment Recovery after Stroke: A Mixture Model,” Ann. Neurol., Vol.87, No.3, pp. 383-393, 2020.
- [61] S. K. Subramanian, A. G. Feldman, and M. F. Levin, “Spasticity may obscure motor learning ability after stroke,” J. Neurophysiol., Vol.119, No.1, pp. 5-20, 2018.
- [62] M. Chancel, C. Brun, A. Kavounoudias, and M. Guerraz, “The kinaesthetic mirror illusion: How much does the mirror matter?,” Exp. Brain Res., Vol.234, No.6, pp. 1459-1468, 2016.
- [63] D. Pérez-Cruzado, J. A. Merchán-Baeza, M. González-Sánchez, and A. I. Cuesta-Vargas, “Systematic review of mirror therapy compared with conventional rehabilitation in upper extremity function in stroke survivors,” Aust. Occup. Ther. J., Vol.64, No.2, pp. 91-112, 2017.
- [64] G. Yavuzer, R. Selles, N. Sezer, S. Sütbeyaz, J. B. Bussmann, F. Köseoğlu, M. B. Atay, and H. J. Stam, “Mirror Therapy Improves Hand Function in Subacute Stroke: A Randomized Controlled Trial,” Arch. Phys. Med. Rehabil., Vol.89, No.3, pp. 393-398, 2008.
 This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.
This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.