Letter:
Development of Microdevices Combining Machine and Life Systems
Yo Tanaka
Center for Biosystems Dynamics Research (BDR), RIKEN
1-3 Yamadaoka, Suita, Osaka 565-0871, Japan
A number of recent studies have exploited the sizes and functional properties of microdevices and cellular mechanical components to construct bio-microdevices. As the scale of microdevices can accommodate different cell sizes and processing capabilities, a number of efficient bioreactors and bioassay systems using cellular functions have been produced. To date, the main focus of these devices has been the analysis of cellular chemical functions. On the other hand, our concept is to use cells as components of devices for fluidic control. To date, various devices have been developed that exploit cellular mechanical functions. The working principle of these devices is novel because they only use chemical energy inputs. In this letter, the recent progress of this study and its characteristics are reviewed.
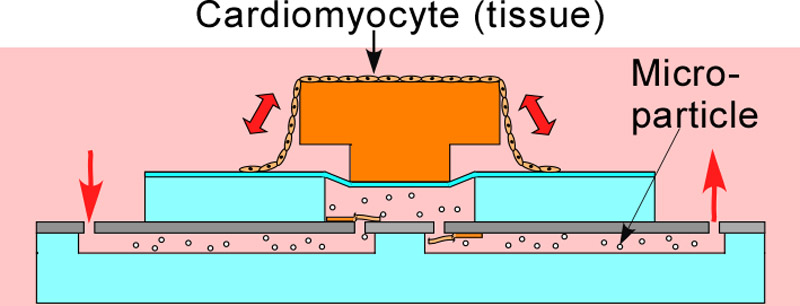
Design and principle of a cardiomyocyte pump
- [1] D. E. W. Patabadige, S. Jia, J. Sibbitts, J. Sadeghi, K. Sellens, and C. T. Culbertson, “Micro Total Analysis Systems: Fundamental advances and applications,” Anal. Chem., Vol.88, Issue 1, pp. 320-338, 2016.
- [2] A. A. M. Faudzi, Y. Sabzehmeidani, and K. Suzumori, “Application of micro-electro-mechanical systems (mems) as sensors: A review,” J. Robot. Mechatron., Vol.32, No.2, pp. 333-343, 2020.
- [3] A. W. Feinberg, “Biological soft robotics,” Annu. Rev. Biomed. Eng., Vol.17, pp. 243-265, 2015.
- [4] J. C. Nawroth, H. Lee, A. W. Feinberg, C. M. Ripplinger, M. L. McCain, A. Grosberg, J. O. Dabiri, and K. K. Parker, “A tissue-engineered jellyfish with biomimetic propulsion,” Nat. Biotechnol., Vol.30, pp. 792-797, 2012.
- [5] Y. Tanaka, K. Morishima, T. Shimizu, A. Kikuchi, M. Yamato, T. Okano, and T. Kitamori, “Demonstration of a PDMS-based bio-microactuator using cultured cardiomyocytes to drive polymer micropillars,” Lab on a Chip, Vol.6, No.2, pp. 230-235, 2006.
- [6] Y. Tanaka, K. Sato, T. Shimizu, M. Yamato, T. Okano, I. Manabe, R. Nagai, and T. Kitamori, “Demonstration of a bio-microactuator powered by vascular smooth muscle cells coupled to polymer micropillars,” Lab on a Chip, Vol.8, No.1, pp. 58-61, 2008.
- [7] Y. Tanaka, K. Morishima, T. Shimizu, A. Kikuchi, M. Yamato, T. Okano, and T. Kitamori, “An actuated pump on-chip powered by cultured cardiomyocytes,” Lab on a Chip, Vol.6, No.3, pp. 362-368, 2006.
- [8] Y. Tanaka, “Electric actuating valves incorporated into an all glass-based microchip exploiting the flexibility of ultra thin glass,” RSC Adv., Vol.3, pp. 10213-10220, 2013.
- [9] Y. Tanaka, K. Sato, and T. Kitamori, “Assembly and simple demonstration of a micropump installing PDMS-based thin membranes as flexible micro check valves,” J. Biomed. Nanotechnol., Vol.5, pp. 516-520, 2009.
- [10] Y. Tanaka, Y. Yanagisawa, and T. Kitamori, “Fluid actuation for a bio-micropump powered by previously frozen cardiomyocytes directly seeded on a diagonally stretched thin membrane,” Sens. Actuators B-Chem., Vol.156, pp. 494-498, 2011.
- [11] Y. Tanaka and H. Fujita, “Fluid driving system for a micropump by differentiating iPS cells into cardiomyocytes on a tent-like structure,” Sens. Actuators B-Chem., Vol.210, pp. 267-272, 2015.
- [12] N. Tanaka, T. Yamashita, Y. Yalikun, S. Amaya, A. Sato, V. Vogel, and Y. Tanaka, “An ultra-small fluid oscillation unit for pumping driven by self-organized three-dimensional bridging of pulsatile cardiomyocytes on elastic micro-piers,” Sens. Actuators B-Chem., Vol.293, pp. 256-264, 2019.
- [13] M. Abulaiti, Y. Yalikun, K. Murata, A. Sato, M. M. Sami, Y. Sasaki, Y. Fujiwara, K. Minatoya, Y. Shiba, Y. Tanaka, and H. Masumoto, “Establishment of a heart-on-a-chip microdevice based on human iPS cells for the evaluation of human heart tissue function,” Sci. Rep., Vol.10, 19201, 2020.
- [14] Y. Aishan, Y. Yalikun, Y. Shen, Y. Yuan, S. Amaya, T. Okutaki, A. Osaki, S. Maeda, and Y. Tanaka, “A chemical micropump actuated by self-oscillating polymer gel,” Sens. Actuators B-Chem., Vol.337, 129769, 2021.
- [15] Y. Tanaka, Y. Noguchi, Y. Yalikun, and N. Kamamichi, “Earthworm muscle driven bio-micropump,” Sens. Actuators B-Chem., Vol.242, pp. 1186-1192, 2017.
- [16] Y. Tanaka, S. Funano, Y. Noguchi, Y. Yalikun, and N. Kamamichi, “A valve powered by earthworm muscle with both electrical and 100% chemical control,” Sci. Rep., Vol.9, 8042, 2019.
- [17] Y. Tanaka, S. Funano, Y. Nishizawa, N. Kamamichi, M. Nishinaka, and T. Kitamori, “An electric generator using living Torpedo electric organs controlled by fluid pressure-based alternative nervous systems,” Sci. Rep., Vol.6, 25899, 2016.
- [18] N. Sasaki, M. Shinjo, S. Hirakawa, M. Nishinaka, Y. Tanaka, K. Mawatari, T. Kitamori, and K. Sato, “A palmtop-sized microfluidic cell culture system driven by a miniaturized infusion pump,” Electrophoresis, Vol.33, pp. 1729-1735, 2012.
- [19] Y. Tanaka, T. Fujikawa, Y. Kazoe, and T. Kitamori, “An active valve incorporated into a microchip using a high strain electroactive polymer,” Sens. Actuators B-Chem., Vol.184, pp. 163-169, 2013.
- [20] Y. Yalikun and Y. Tanaka, “Ultra-thin glass sheet integrated transparent diaphragm pressure transducer,” Sens. Actuators A-Phys., Vol.263, pp. 102-111, 2017.
- [21] Y. Yuan, Y. Yalikun, S. Amaya, Y. Aishan, Y. Shen, and Y. Tanaka, “Fabrication of ultra-thin glass sheet by weight-controlled load-assisted precise thermal stretching,” Sens. Actuators A-Phys., Vol.321, 112604, 2021.
 This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.
This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.