Paper:
Development of Artificial Skin Using Keratin Film for Evaluation of Puncture Performance of Microneedle
Ryo Nishino*1, Seiji Aoyagi*1, Masato Suzuki*1, Atsushi Ueda*1, Yuki Okumura*1, Tomokazu Takahashi*1, Ryota Hosomi*1, Kenji Fukunaga*1, Daisuke Uta*2, Tomonori Takazawa*3, and Toshihiro Fujii*4
*1Kansai University
3-3-35 Yamate-cho, Suita, Osaka 564-8680, Japan
*2University of Toyama
2630 Sugitani, Toyama-shi, Toyama 930-0194, Japan
*3Gunma University
3-39-15 Showa-machi, Maebashi, Gunma 371-8511, Japan
*4Shinsyu University
3-15-1 Tokida, Ueda, Nagano 386-8567, Japan
Humans do not feel pain when bitten by mosquitos; therefore, we have attempted to develop a microneedle that mimics the puncturing mechanism of mosquitos. We have quantitatively evaluated the puncturing performance of the developed microneedle by puncturing an artificial skin made from polydimethylsiloxane (PDMS), a kind of silicon rubber. Unlike the mono-layered PDMS, however, animal skin including human skin is structured to have a hard stratum corneum, epidermis and dermis over soft subcutaneous tissue. In this paper, we propose an artificial skin having a two-layered hard/soft structure, constructed from PDMS with a human-hair-derived keratin film adhered onto the top surface. We evaluated the hardness of the keratin film (Young’s modulus) and found that it could qualitatively simulate the hard layers of the skin including the stratum corneum. The artificial skin we developed reproduced the following phenomena: the decrease in resistance force of animal skin at the point when the needle penetrates the surface followed by variation in resistance due to the stick-slip phenomenon as the needle penetrates more deeply.
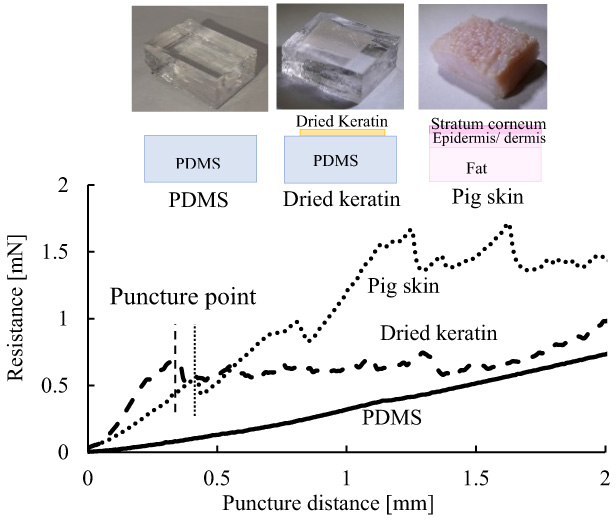
Keratin film reproduces puncture and stick-slip of pig skin
- [1] S. Aoyagi, H. Izumi, and M. Fukuda, “Biodegradable Polymer Needle with Various Tip Angles and Consideration on Insertion Mechanism of Mosquito’s Proboscis,” Sensors and Actuators, Vol.A143/1, pp. 20-28, 2008.
- [2] H. Izumi and S. Aoyagi, “Novel Fabrication Method for Long Silicon Microneedles with Three-Dimensional Sharp Tips and Complicated Shank Shapes by Isotropic Dry Etching,” Trans. on Electrical and Electronic Engineering, Vol.2, No.3, pp. 328-334, 2007.
- [3] S. Aoyagi, “Visualization in Development of Microneedle – Development of Painless Needle Imitating Mosquito –,” J. of the Visualization Society of Japan, Vol.33, No.131, pp. 145-148, 2013 (in Japanese).
- [4] H. Kitada, H. Yamamoto, S. Yamamoto, M. Suzuki, S. Aoyagi, T. Takahashi, K. Fukunaga, R. Hosomi, T. Takazawa, D. Uta, Y. Kawajiri, K. Nakayama, and T. Hikitsuchi, “Observation of Mosquito Skin Penetration and Blood Suction in laboratory animals,” Proc. of 2017 JSPE Autumn Meeting, N-18, pp. 897-898, 2017 (in Japanese).
- [5] A. N. Clement, “The Biology of Mosquitoes,” CABI Publishing, pp. 224-234, 2000.
- [6] S. Aoyagi, “Overview of Microneedles,” J. of the Japan Society for Precision Engineering, Vol.82, No.12, pp. 999-1004, 2016 (in Japanese).
- [7] M. Suzuki et al., “Ultrafine three-dimensional (3D) laser lithographic fabrication of microneedle and its application to painless insertion and blood sampling inspired by mosquito,” 2015 IEEE/RSJ Int. Conf. on Intelligent Robots and Systems (IROS), 2015.
- [8] Y. Hara, M. Yamada, C. Tatsukawa, T. Takahashi, M. Suzuki, and S. Aoyagi, “Fabrication of Stainless Steel Microneedle with Laser-Cut Sharp Tip and its Penetration and Blood Sampling Performance,” Int. J. Automation Technol., Vol.10, No.6, pp. 950-957, 2016.
- [9] Y. Hara, M. Yamada, C. Tatsukawa, T. Takahashi, M. Suzuki, and S. Aoyagi, “Laser Fabrication of Jagged-Shaped Stainless Steel Microneedle Imitating Mosquito’s Maxilla,” Int. J. Automation Technol., Vol.10, No.6, pp. 958-964, 2016.
- [10] S. Terashima et al., “Fabrication of microneedle using poly lactic acid sheets by thermal nanoimprint,” Precision Engineering, Vol.59, pp. 110-119, 2019.
- [11] M. Suzuki et al., “Fabrication of microneedles precisely imitating mosquito’s proboscis by nanoscale tree dimensional laser lithography and its characterization,” Proc. of 2015 18th Int. Conf. on Solid-State Sensors, Actuators and Microsystems (Transducers), pp. 121-124, 2015.
- [12] S. A. Ranamukhaarachchi et al., “A micromechanical comparison of human and porcine skin before and after preservation by freezing for medical device development,” Scientific Reports, Vol.6, 32074, 2016.
- [13] M. Ogura et al., “Development of puncture Jig for preventing from buckling of microneedle imitating labium of mosquito,” Symp. on Micro-Nano Science and Technology, pp. 233-234, 2013 (in Japanese).
- [14] P. G. Agache et al., “Mechanical properties and Young’s modulus of human skin in vivo,” Archives of Dermatological Research, Vol.269, pp. 221-232, 1980.
- [15] F. M. Hendriks et al., “A numerical-experimental method to characterize the non-linear mechanical behaviour of human skin,” Skin Research and Technology, Vol.9, No.3, pp. 274-283, 2003.
- [16] J. F. Manschot and A. J. Brakkee, “The measurement and modelling of the mechanical properties of human skin in vivo – II. The model,” J. of Biomechanics, Vol.19, No.7, pp. 517-521, 1986.
- [17] M. Suzuki, T. Takahashi, and S. Aoyagi, “3D laser lithographic fabrication of hollow microneedle mimicking mosquitos and its characterization,” Int. J. Nanotechnology, Vol.15, No.1, pp. 157-173, 2018.
- [18] M. Yamada, T. Takahashi, M. Suzuki, S. Aoyagi, R. Hosomi, K. Fukunaga, D. Uta, and T. Takazawa, “Proposal of new blood sampling method using microneedle – use of reciprocating rotation, visualization of blood vessels –, ” Proc. Spring Meeting of Japan Soc. Prec. Eng., pp. 531-532, 2018 (in Japanese).
- [19] S. Aoyagi, H. Izumi, and M. Fukuda, “Biodegradable Polymer Needle with Various Tip Angles and Consideration on Insertion Mechanism of Mosquito’s Proboscis,” Sensors and Actuators, Vol.A143, No.1, pp. 20-28, 2008.
- [20] H. Izumi, M. Suzuki, S. Aoyagi, and T. Kanzaki, “Realistic imitation of mosquito’s proboscis: Electrochemically etched sharp and jagged needles and their cooperative inserting motion,” Sensors and Actuators, Vol.A165, No.1, pp. 115-123, 2011.
- [21] S. Aoyagi, Y. Takaoki, H. Takayanagi, C.-H. Huang, T. Tanaka, M. Suzuki, T. Takahashi, T. Kanzaki, and T. Matsumoto, “Equivalent Negative Stiffness Mechanism Using Three Bundled Needles Inspired by Mosquito for Achieving Easy Insertion,” Proc. IEEE/RSJ Int. Conf. Intelligent Robots and Systems (IROS), pp. 2295-2300, 2012.
- [22] N. D. M. Begg and A. H. Slocum, “Audible frequency vibration of puncture-access medical devices,” Medical Eng. Physics, Vol.36, No.3, pp. 371-377, 2014.
- [23] S. Henry, D. V. McAllister, M. G. Allen, and M. R. Prausnitz, “Micromachined Needles for the Transdermal Delivery of Drugs,” Proc. MEMS’98, pp. 494-498, 1998.
- [24] T. Matsumoto, “Skin biomechanics from microscopic viewpoint: mechanical properties and their measurement of horny layer, living epidermis, and dermis,” Fragrance J., Vol.35, No.2, pp. 36-40, 2017 (in Japanese).
- [25] J. C. J. Wei et al., “Allometric scaling of skin thickness, elasticity, viscoelasticity to mass for micro-medical device translation: from mice, rats, rabbits, pigs to humans,” Scientific Reports, Vol.7, No.1, 15885, 2017.
- [26] T. Fujii and Y. Ide, “Preparation of translucent and flexible human hair protein films and their properties,” Biological and Pharmaceutical Bulletin, Vol.27, No.9, pp. 1433-1436, 2004.
- [27] T. Fujii, S. Takayama, and Y. Ito, “A novel purification procedure for keratin-associated proteins and keratin from human hair,” J. of Biological Macromolecules, Vol.13, No.3, pp. 92-106, 2013.
- [28] T. Fujii et al., “Preparation of Model Shampoos and Identification of the Component to Reduce the Friction Coefficient Using the Keratin Film,” J. of Fiber Science and Technology, Vol.72, No.8, pp. 166-171, 2016.
- [29] T. Fukui et al., “In vivo mechanical properties of thoracic aortic aneurysmal wall estimated from in vitro biaxial tensile test,” Bio-medical Materials and Engineering, Vol.15, No.4, pp. 295-305, 2005.
- [30] A. Ullah, C. M. Kim, and G. M. Kim, “Porous polymer coatings on metal microneedles for enhanced drug delivery,” Royal Society Open Science, Vol.5, No.4, 171609, 2018.
 This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.
This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.