Research Paper:
Effectiveness of Air Classification as a Pretreatment for Chromium Separation from Electric Arc Furnace Slag
Masami Koide*1,*2, Keishi Oyama*3
 , Yutaro Takaya*4,*5
, Yutaro Takaya*4,*5
 , and Chiharu Tokoro*4,*5,†
, and Chiharu Tokoro*4,*5,†

*1Production Planning and Coordination Department, Kyoei Steel Ltd.
1-4-16 Dojimahama, Kita-ku, Osaka 530-0004, Japan
*2Graduate School of Creative Science and Engineering, Waseda University
Tokyo, Japan
*3Department of Earth Resources Engineering, Kyushu University
Fukuoka, Japan
*4Faculty of Science and Engineering, Waseda University
Tokyo, Japan
*5Faculty of Engineering, The University of Tokyo
Tokyo, Japan
†Corresponding author
Electric arc furnace slag is a byproduct of the steel scrap refining process. It must be effectively used as a recycled resource because of its enormous production volume. Cement usage is a promising application; however, there is a risk that chromium (Cr) in the slag will be oxidized to hexavalent chromium during the cement manufacturing process and subsequently leach into the environment. Because chemical treatments, such as alkali roasting, are required to significantly reduce Cr in electric arc furnace slag, it is important to establish a chemical-free physical pretreatment to reduce the overall energy demand. In this study, we investigated the presence of Cr compounds in oxidizing and reducing slag and comparatively evaluated the effectiveness of physical pretreatment using air classification. The results showed that Cr mainly existed as a spinel phase bonded to other metals, such as Fe and Mg, and this feature was common in both slags. In air classification experiments using an elbow-jet air classifier, the Cr concentrations were similar for all classifications of the oxidizing slag. However, the Cr concentrations in the reducing slag were higher in the coarse powder fraction, whereas Ca tended to be concentrated in the fine powder fraction. Air classification using an elbow-jet air classifier was effective in reducing the Cr content in the coarse fraction of reducing slag, suggesting that it could be used as a pretreatment prior to chemical processing to reduce the overall energy demand. The contrasting classification behavior of oxidizing and reducing slags highlights the importance of slag-type-specific separation strategies.
1. Introduction
Steel is an essential material in modern society and is utilized in various fields, such as construction, transportation, and manufacturing. In 2022, the global crude steel production reached 1,887,809 thousand tons 1. Recently, efforts to achieve carbon neutrality on a global scale have accelerated 2, and the steel industry, which is a major source of greenhouse gas emissions, is required to reduce its carbon dioxide emissions. The electric arc furnace method, which uses steel scrap as a raw material, emits only approximately one-fourth of the carbon dioxide emitted by the blast furnace method. Therefore, it will play a crucial role in the decarbonization of steel manufacturing processes in the future. Approximately 120 kg of electric arc furnace slag is produced per ton of crude steel. In Japan, the production volume in 2023 is 2,735 thousand tons 3. Although these slags are effectively recycled, their composition varies with each production lot because of impurities in the steel scrap and refining additives. Consequently, for safety and environmental reasons, their use is primarily restricted to roadbed and civil engineering materials. Because the demand for these applications is heavily influenced by regional construction needs, it is essential to broaden the applications of electric arc furnace slag to ensure its stable and sustainable utilization in a resource-circulating society.
Electric arc furnace slag mainly consists of oxides such as calcium (Ca), silicon (Si), iron (Fe), and aluminum (Al). When considering its use as a raw material for cement, there is a strong concern that trivalent chromium (Cr(III)) in the slag may be oxidized to hexavalent chromium (Cr(VI)) during the cement manufacturing process and leach out after the product is formed. Cr(VI) is highly toxic and carcinogenic, causing health hazards and environmental pollution. Therefore, to safely use electric arc furnace slag as a raw material for cement, it is important to separate and reduce the concentration of Cr in advance. Regarding the concentration of Cr in cement, the guidelines established by the Japan Cement Association require that the concentration of water-soluble Cr(VI) should not exceed 20 mg/kg 4. On the other hand, there are no clearly defined standards for the concentration of Cr in cement raw materials, and each cement manufacturer presents acceptance criteria to their raw material suppliers. The concentration is generally expected to range from 0.01–0.1 wt%, with a minimal Cr content being desirable.
To significantly reduce the concentration of Cr in electric arc furnace slag, chemical treatments such as acid or alkali leaching and alkali roasting are required 5,6,7. However, these methods require substantial energy and chemicals. Therefore, introducing a physical pretreatment to concentrate and separate Cr before these chemical treatments is an important strategy for reducing both environmental impact and cost 8,9. Physical separation techniques for separating Cr from electric arc furnace slag have been primarily studied with the aim of utilizing it as a secondary resource 10,11. Although magnetic and gravity separation have been attempted, the effectiveness of Cr separation varies depending on the slag type and separation method. It is important to employ methods that are appropriate for the specific chemical form of Cr in the target slag. Morphological observations and elemental analyses of electric arc furnace slag cross-sections using scanning electron microscopy-energy dispersive X-ray spectroscopy (SEM-EDS) confirmed the presence of phases with high Cr content. Among them, the size of Cr-containing phases in carbon steel slag tends to be less than approximately 100 μm [10–13]. This study focuses on this point and investigates the selective separation of Cr contained in electric arc furnace slag through classification.
Izumi et al. conducted air classification using an elbow-jet air classifier on crushed solar panel sheets as an example of multi-component crushed materials 14. The results of the three-point classification experiments using the same classifier quantitatively clarified that the classification behavior was influenced not only by the difference in particle size of each component but also by the difference in specific gravity. Given that the electric arc furnace slag in this study contained various elements, classification by the elbow-jet air classifier was expected to depend on both the particle size and specific gravity. In this study, we investigated the possibility of separating Cr from electric arc furnace slag using an elbow-jet air classifier, which is a type of air classifier, as one of the physical separation methods that considers differences in particle size and specific gravity. By focusing on the differences in the properties of oxidizing and reducing slag and comparing the classification behavior of Cr, we evaluated the applicability and limitations of physical pretreatment.

Fig. 1. Photographs of (a) oxidizing and (b) reducing slag produced by an electric arc furnace.
2. Materials and Methods
2.1. Electric Arc Furnace Slag
In this study, two types of electric arc furnace slag generated during the manufacturing process of carbon steel were used. The appearances of both slags are shown in Fig. 1. The first is oxidizing slag produced during the oxidizing refining process in the electric arc furnace, and the second is reducing slag produced during the reducing refining process in the ladle furnace. The oxidizing slag consisted of blackish particles with a diameter of approximately 30 mm. The reducing slag is generally a white powder containing black and white particles. Approximately 1 kg of both slag samples were collected from the cooling pits of a Japanese steel manufacturer’s storage facility. To confirm the representativeness of the samples, the composition trends were compared with the average values of the X-ray fluorescence (XRF) analysis results (Table 1) from approximately 30 samples collected over the past two years. It was confirmed that the composition trends were generally similar.
Table 1. Chemical composition of oxidizing and reducing slag determined by XRF analysis [wt%].

2.2. Characterization
To investigate the form in which Cr exists in electric arc furnace slag, both oxidizing and reducing slag were subjected to SEM-EDS and X-ray absorption fine structure (XAFS) analyses. For the SEM-EDS analysis, slag particles of approximately 15 mm were embedded in resin and polished to prepare cross-sections. For the reducing slag, the observations were focused on relatively large particles that could be cut and polished. Using tabletop microscopes (Hitachi High-Tech, TM3030Plus and TM4000Plus), reflected electron images were observed at a magnification of 1,000 times. Additionally, mapping and spot analyses were conducted to determine the distribution of each element and perform localized elemental analyses of the crystal grains. For XAFS analysis, both slags were ground using a vibration mill (CMT Co., Ltd., TI-100), and the X-ray absorption near edge structure (XANES) spectra at the Cr-K absorption edge were measured using the fluorescence method. Standard samples were measured using the transmission method. This analysis was conducted at BL11S2 and BL5S1 of the Aichi Synchrotron Radiation Center, and data analysis was performed using the Athena module within the Demeter software package 15.
Table 2. Conditions for ball milling of oxidizing slag.

2.3. Experimental Methods
2.3.1. Sample Preparation
For both slag samples, approximately 100 g of the 1 kg collected from the site was used for the elbow-jet air classification experiments. As the oxidizing slag had a particle size of approximately 30 mm when collected from the site, it was crushed before the separation experiment. The slag was crushed using a jaw crusher (Retsch, BB50) at a rotation speed of 600 rpm and a gap width of 2 mm, followed by further grinding with a ball mill (Sato Seisakusho) under the conditions listed in Table 2. The reducing slag was in powder form when collected from the site; therefore, no grinding was performed. Approximately 100 g of each slag sample was divided, and the particle size distribution was determined and used for the elbow-jet air classification experiments.
2.3.2. Particle Size Distribution
To determine the particle size distribution, both slag samples were classified using 13 different sieves with mesh sizes of 32, 45, 63, 90, 125, 180, 250, 350, 500, 710, 1,000, 1,400, and 2,000 μm. The weights of each of the 14 classified fractions were measured.
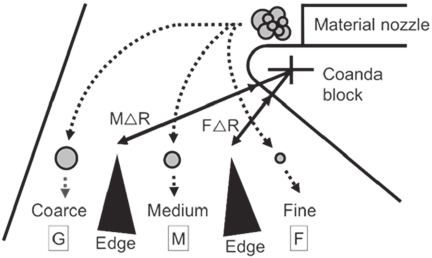
Fig. 2. Schematic of the elbow-jet classifier and image of the three-point classification.
Table 3. Conditions for elbow-jet air classification.

2.3.3. Elbow-Jet Air Classification Experiment
The elbow-jet air classifier is a dry fine powder classifier that utilizes the difference in the inertial force acting on particles and the air resistance received from the airflow to enable simultaneous three-point classification. The applicable particle size range is 0.5–50 μm, making it suitable for classifying materials such as toner and battery components. Owing to its simple internal structure, it can be scaled up while maintaining its performance, and commercial-scale versions have already been realized. Fig. 2 shows a schematic of the device and the concept of three-point classification. When the raw powder is fed into the device, the raw powder and feed air are simultaneously ejected from the raw material supply nozzle 16. The jet stream tends to follow the Coanda block owing to the Coanda effect, which is the tendency of a jet to adhere to a nearby surface 17. Particles with larger sizes or specific gravities possess greater inertia and travel farther, whereas those with smaller sizes or specific gravities have less inertia and tend to follow the Coanda block. Owing to this difference in flight trajectory, larger or heavier particles are classified as coarse powders, whereas smaller or lighter particles are classified as fine powders. Previous studies have reported that Cr-containing phases in electric arc furnace slag are clearly separated from the matrix phase of silicates containing Ca 10,12. Because Cr is expected to have a relatively high specific gravity among the elements constituting electric arc furnace slag, it is anticipated that classification using this device will efficiently separate Cr on the coarse powder side. Both slags were subjected to an elbow-jet air classifier (Nittetsu Mining Co., Ltd., EJ-L-3) and classified into coarse, medium, and fine powders. The classification conditions are listed in Table 3. The classification points were set by trial and error to ensure that the weight of each classified fraction was sufficient for subsequent XRF analysis, considering the size of the Cr-containing phases in the sample. The slag samples were subjected to the classifier, and the fractions obtained from the three-point classification were ground using a vibration mill, and XRF analysis was performed using a handheld X-ray fluorescence analyzer (EVIDENT, VMR-CCC-G3-J).
3. Results and Discussion
3.1. Form of Cr in Electric Arc Furnace Slag
3.1.1. Morphological Observation and Elemental Distribution of Cr-Containing Phases

Fig. 3. SEM images of (a) oxidizing and (b) reducing slag.

Fig. 4. SEM-EDS elemental mapping of oxidizing slag.
Figure 3 shows the SEM images of the oxidizing and reducing slag. Observations of the reflected electron images revealed that the oxidizing slag contained relatively large amounts of various crystal grains throughout the entire field of view. In contrast, the reducing slag generally had smaller crystal grains, with some grown crystal grains scattered within a uniform phase.
Elemental mapping analysis was performed to confirm the distribution of Cr and the elements coexisting with Cr. A mapping image of the oxidizing slag is shown in Fig. 4. Cr-rich particles with sizes of up to approximately 50 μm were identified. The distributions of Fe, Al, and Mg overlapped with the Cr-rich phases, indicating that these elements coexisted with Cr. Notably, Al was often observed to coexist with Cr and surround Cr-rich particles. These particles were commonly observed across many fields of view in the slag cross-section. Other observations included the presence of relatively large metallic Fe phases and oxide phases containing Ca and Si, which appeared to form the matrix phase.

Fig. 5. SEM-EDS elemental mapping of reducing slag.
The mapping image of the reducing slag is shown in Fig. 5. In the reducing slag, particles with high concentrations of Cr were also identified, with sizes of approximately 40 μm. The distributions of Fe and Al overlapped with these Cr-rich phases, indicating their coexistence with Cr. Unlike the oxidizing slag, the surrounding areas of the dispersed crystal grains in the reducing slag exhibited limited crystal growth, and large metallic Fe phases similar to those found in the oxidizing slag were rarely observed. The oxide phases containing Ca and Si were confirmed to exist as the matrix phase, similar to that of the oxidizing slag.
To investigate the Cr concentration in the Cr-rich crystal grains, spot elemental analysis was performed at the locations marked with crosses in Fig. 3. The Cr concentrations in the oxidizing and reducing slag were 15.50 and 7.73 wt%, respectively. Previous studies that conducted SEM-EDS analysis on electric arc furnace slag and synthetic slag collected from the site reported that the distribution of Cr in the slag overlaps with Fe, Al, Mg, and Mn, whereas Ca and Si are distributed in different locations 18,19,20. This study confirmed similar trends to those reported previously. In particular, the morphology of the particles in the oxidizing slag closely resembled the Cr-containing phases observed in previous studies. Zeng et al. 19 synthesized slag with an FeO concentration of 20 mass% and analyzed it using SEM-EDS after air cooling. They reported that Cr was most concentrated at the center of the spinel phase, with Fe surrounding it. However, in this study, Al was found to surround the Cr-rich particles in the oxidizing slag. It has been reported that the elements that dissolve in the Cr-containing spinel phase vary depending on the slag composition, basicity, and cooling rate 19,21,22,23. Therefore, the basicity and cooling rate of the oxidizing slag used in this study also influenced the phase formation.
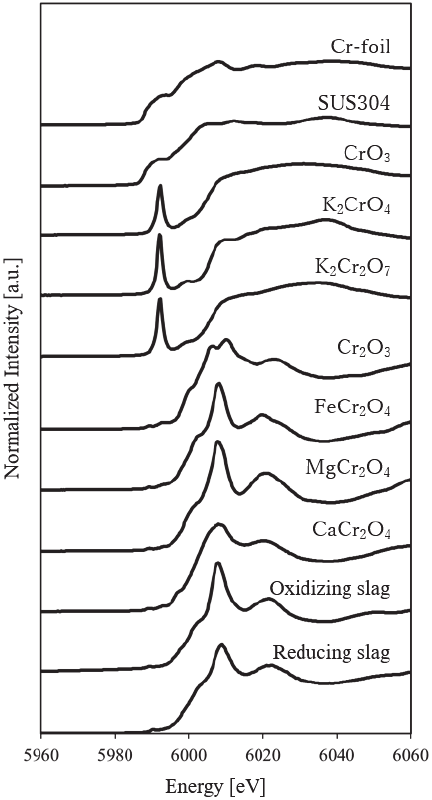
Fig. 6. XAFS spectra of standard samples (Cr-foil, SUS304, CrO\(_3\), K\(_2\)CrO\(_4\), K\(_2\)CrO\(_7\), Cr\(_2\)O\(_3\), FeCr\(_2\)O\(_4\), MgCr\(_2\)O\(_4\), and CaCr\(_2\)O\(_4\)) and slag samples (oxidizing and reducing slag).
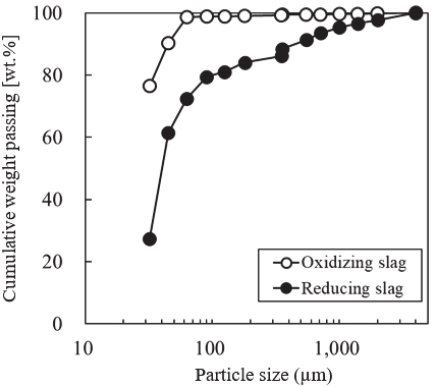
Fig. 7. The particle size distributions of the oxidizing and reducing slags used for air classification.
Table 4. Chemical composition of coarse, medium, and fine-size fractions determined by XRF analysis [wt%].

3.1.2. Investigation of Chromium Valence and Cr-Containing Phases
The characteristics of chromium observed by SEM-EDS were consistent with the Cr-containing phases identified in previous studies. According to XRD analysis conducted in prior research, these phases are reported to be spinel-type phases such as MgCr\(_2\)O\(_4\) and FeCr\(_2\)O\(_4\). Furthermore, spinel phases are oxides containing divalent and trivalent metals, and it has been confirmed that during the cooling process, some metals are substituted to form phases such as (Mg, Fe)(Cr, Fe)\(_2\)O\(_4\) and (Mg, Mn)(Cr, Al, Fe)\(_2\)O\(_4\) 19,24,25,26.
In this study, it was assumed that the slag samples contained these spinel phases. However, owing to the difficulty in detecting Cr-containing phases by XRD analysis from the perspectives of Cr concentration and crystallinity, XAFS analysis was attempted to investigate the valence and mineral phases of Cr, which is capable of analyzing substances with low concentration and low crystallinity.
The XAFS spectra of various Cr compounds and slag samples are shown in Fig. 6. In both the oxidizing and reducing slag, the characteristic pre-edge peaks observed in hexavalent Cr standard samples, such as CrO\(_3\), K\(_2\)CrO\(_4\), and K\(_2\)Cr\(_2\)O\(_7\), were not detected. The absorption edge positions were approximately the same as those of trivalent Cr standard samples such as Cr\(_2\)O\(_3\) and FeCr\(_2\)O\(_4\), confirming that the Cr present in both slags was trivalent.
Furthermore, the spectra of both slags were more similar to those of spinel-type oxides, such as FeCr\(_2\)O\(_4\), MgCr\(_2\)O\(_4\), and CaCr\(_2\)O\(_4\), than to that of the simple oxide Cr\(_2\)O\(_3\). This indicates that Cr exists as spinel-type oxides in both slags. Focusing on the peaks near the absorption edge, the oxidizing slag showed peak heights similar to those of FeCr\(_2\)O\(_4\) and MgCr\(_2\)O\(_4\), while the reducing slag exhibited a shape close to that of CaCr\(_2\)O\(_4\). These results suggest that FeCr\(_2\)O\(_4\) and MgCr\(_2\)O\(_4\) are present in relatively high proportions in the oxidizing slag. Moreover, in the reduced slag, Cr is considered to be primarily incorporated into the CaCr\(_2\)O\(_4\) phase. Although the EDS mapping (Fig. 5) seems to indicate an inverse distribution between Ca and Cr, this is likely due to the extremely high Ca concentration in the reduced slag (33.12 wt%), which makes the Ca concentration within the CaCr\(_2\)O\(_4\) particles appear relatively lower than that of the surrounding matrix.
Zhao et al. demonstrated that the formation of CaCr\(_2\)O\(_4\) is promoted when a high CaO content is present in the steel slag 23. Therefore, it is reasonable to assume that CaCr\(_2\)O\(_4\) is present in the reducing slag, which has a relatively high Ca concentration. However, it is difficult to fully explain the spectra of both slags using only these three standard samples. The presence of other elements, such as Al, and the crystallinity of the slag also influence the spectra.
3.2. Separation of Chromium by Air Classification
3.2.1. Particle Size Distribution of Electric Arc Furnace Slag
The particle size distributions of both slag samples used in the elbow-jet classification experiment are shown in Fig. 7. The applicable particle size range for the elbow-jet classifier was 0.5–50 μm. Among the slag samples used in this study, the weight percentage of particles with a diameter of 45 μm or less was 90.3 wt% for oxidizing slag and 61.4 wt% for reducing slag, respectively.
3.2.2. Separation of Chromium by Air Classification
The weights and XRF analysis results of the slag samples subjected to the classifier and three-point classified products are listed in Table 4. LE (light element) represents the combined detection values of light elements from Li to Na. The weights of the coarse, medium, and fine powders for the oxidizing slag were 12.80 g (57.42 wt% of the total classified products), 4.82 g (21.62 wt%), and 4.67 g (20.95 wt%), respectively. For the reducing slag, the weights were 12.51 g (47.42 wt%), 8.61 g (32.64 wt%), and 5.26 g (19.94 wt%), respectively.
The Cr concentration of the oxidizing slag sample subjected to classification was 2.05 wt%. The Cr concentration in the classified products was 2.02 wt% for the coarse powder, 2.29 wt% for the medium powder, and 1.92 wt% for the fine powder, showing no significant changes. In contrast, the Cr concentration of the reducing slag sample subjected to classification was 0.25 wt%. The Cr concentrations in the classified products were 0.46 wt% for the coarse powder, 0.15 wt% for the medium powder, and 0.07 wt% for the fine powder, indicating a higher concentration in the coarse powder.
The Cr distribution ratios, calculated based on the weight and Cr concentration of each classified product, are shown in Fig. 8. The width of each bar represents the proportion of the total weight of the classified products for each slag, normalized to 100%. In the oxidizing slag, the Cr concentration did not show significant differences among the classified products, and the distribution ratio was approximately proportional to the weight of the classified products. In contrast, in the reducing slag, the Cr concentration was higher in the coarse powder, resulting in a higher distribution ratio despite the lower weight proportion compared to the oxidizing slag. This indicates that the Cr-containing phases in the reducing slag exist as relatively large particles and are preferentially distributed on the coarse powder side. However, even in the fine powder, which exhibited the lowest Cr concentration, the target level of 0.01 wt% was not achieved.
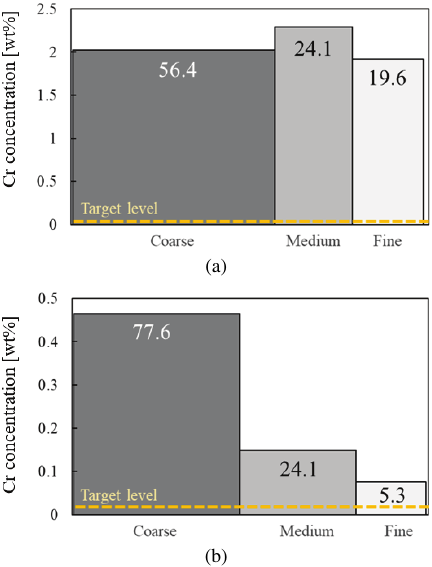
Fig. 8. Cr distribution ratio for each size fraction of (a) oxidizing and (b) reducing slag. The figures shown in the bar charts indicate the mass ratio of the separation fraction.
The pretreatment of oxidizing slag by ball milling was conducted to isolate the Cr-containing phases identified in the previous SEM-EDS analysis. The milling process was performed to achieve a particle size of approximately 50 μm or less. Additionally, to facilitate the preferential removal of relatively softer elements such as Ca from the surface of the slag particles, the rotational speed was maintained at 60 rpm to reduce the collision energy between the balls and the mill walls. However, no significant changes in Cr concentration were observed among the classified products after air classification. Similarly, no clear changes were noted for the other elements between the classified products. This suggests that the ball milling process primarily resulted in simple volumetric grinding without sufficient promotion of phase isolation or selective grinding. Oxidizing slag generally contains more metal oxides, such as Fe, Al, and Mg, than reducing slag. Consequently, it is harder, denser, and possesses wear-resistant physical properties, making it widely used in applications such as concrete aggregates and as road base materials. Therefore, the isolation of Cr-containing phases by ball milling proved to be challenging, and it was inferred that the Cr-containing phases were distributed similarly in both the coarse and fine powders during air classification.
The primary mineral phase constituting the reducing slag is Ca\(_2\)SiO\(_4\), which becomes powdered owing to volumetric expansion during the phase transition in the slow cooling process after slag formation 27,28. The slag samples used in the experiment were initially in lump form but became powdered during the slow cooling process, indicating that this reaction occurred. Therefore, the particles containing Ca and Si in the slag samples were likely to be finely powdered, resulting in relatively smaller particle sizes and being classified into the fine powder side. In contrast, the phase formation of Cr-containing spinel phases is considered to have different characteristics than that of Ca. Zhao et al. investigated the phase formation behavior of various elements, focusing on Cr, by rapidly cooling artificially generated slag from 1600°C, or by cooling it from 1600°C to 1500°C, 1400°C, and 1300°C at a rate of 5°C/min, followed by rapid quenching with water 29. The proportion of Cr in the spinel phase increased as the quenching start temperature decreased. Additionally, the size of the spinel phase tended to increase with lower quenching start temperatures, suggesting that the crystal growth was promoted by slow cooling. The concentrations of Mg, Al, and Fe in the spinel phase varied depending on the quenching temperature but were consistently higher than those in the other phases. Therefore, Cr in the reducing slag samples used in this study was considered to form spinel-type mineral phases combined with these metals. As a result of crystal growth during the cooling process, the particle sizes of these phases became relatively larger than those of the powdered Ca and Si, and they were classified into the coarse powder side.
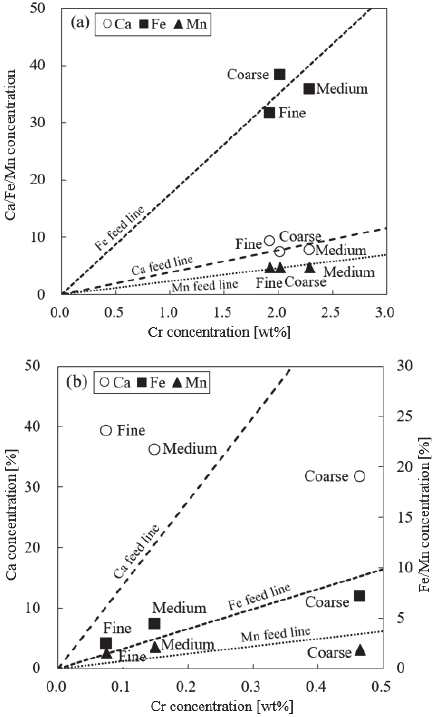
Fig. 9. Relationship between Cr and Ca/Fe/Mn concentrations in each size fraction of (a) oxidizing and (b) reducing slag.
Figure 9 shows the relationship between the Cr concentration and the concentrations of Ca, Fe, and Mn in the classified products. The dotted lines in the figure pass through the points representing the Cr concentration and the concentrations of Ca, Fe, and Mn in the raw slag. When considering the use of electric arc furnace slag as a raw material for cement, it is desirable for the classified products to have a high concentration of Ca, the main component of cement raw materials, and a low concentration of Cr. In the case of the oxidizing slag, although a high concentration of Ca was observed in the fine powder, the plots of the classified products generally clustered around the dotted lines, indicating that the separation between Ca and Cr was not significantly promoted. In contrast, in the case of reducing slag, the plots of the classified products were relatively dispersed. The fine powder exhibited a clear reduction in Cr concentration and an increase in Ca concentration. This trend is favorable for considering the processing of reducing slag for cement raw materials, suggesting that physical separation using an elbow-jet classifier for air classification is effective for Cr separation in reducing slag.
The alkaline roasting-water leaching process is a chemical treatment capable of selectively and accurately recovering Cr from electric arc furnace slag 6,7. However, this process requires a large amount of alkali agents to convert the insoluble Cr-containing spinel phase into soluble compounds 30. This is because, during the high-temperature roasting of the mixture of electric arc furnace slag and alkali agents, the alkali agents are consumed not only in the oxidation reactions of Cr but also in those of Fe and Mn. Consequently, an excessive amount of alkali agents must be added. Therefore, if physical pretreatment can simultaneously reduce Fe and Mn in the Cr-enriched phase, it may be possible to reduce the amount of alkali agent consumed and the energy required for roasting. As shown in Fig. 9, the degree of separation of Fe did not vary significantly between the slags, but the concentration was consistently higher in the coarse powder and lower in the fine powder. In particular, the Fe concentration in the fine powder fraction decreased by 50.61% compared to the feed sample. Typically, electric arc furnace slag contains metallic Fe that is reduced during smelting. Additionally, as previously mentioned, Fe exists as relatively large and dense particles in the spinel phase, which likely led to its concentration in the coarse powder. Similarly, Mn showed lower concentrations in the fine powder, but the plots were close to the dotted lines, indicating a low separation efficiency. Mn is more easily oxidized than Si but less easily than Fe at approximately 1600°C in molten steel. The previously mentioned SEM-EDS mapping analysis confirmed that Mn was distributed throughout the field of view, suggesting that it coexists with multiple elements in an ongoing reaction state. Therefore, it can be inferred that Mn was not concentrated in specific classified products when subjected to the elbow-jet classifier. Thus, considering subsequent chemical treatments, air classification using the elbow-jet classifier could effectively remove not only Cr but also Fe and Mn, which are energy-consuming substances, making it a potentially effective pretreatment method.
4. Conclusions
In this study, an elbow-jet classification experiment was conducted to investigate the potential for separating Cr from electric arc furnace slag using air classification. The Cr concentration in the classified products showed no significant change compared to the pre-classification state for oxidizing slag, whereas for reducing slag, the Cr concentration was higher in the coarse powder and lower in the fine powder. For lump oxidizing slag, pretreatment with ball milling was expected to promote the isolation of Cr-containing phases. However, no substantial changes in elemental concentrations were observed across the classified fractions, suggesting that sufficient phase isolation was not achieved, which reduced the classification efficiency. Regarding the reducing slag, the primary mineral Ca\(_{2}\)SiO\(_{4}\) is known to become powdered during the cooling process after formation, whereas the Cr-containing phases undergo crystal growth and increase in size. Therefore, the particles containing Ca were relatively smaller and concentrated in the fine powder, whereas the particles containing Cr were relatively larger and concentrated in the coarse powder. Additionally, Fe was significantly concentrated in the coarse powder, either as metallic Fe or as part of Cr-containing phases, and Mn exhibited lower concentrations in the fine powder. The Cr concentration exhibited an inverse trend to that of Ca, which is the main component of cement. The simultaneous removal of Fe and Mn, which are highly reactive and consume energy during chemical Cr removal, along with Cr, is advantageous for preparing cement raw materials. This suggests that the fine powder, which is enriched in Ca and depleted in Cr, Fe, and Mn, is effective as a pretreatment for the chemical processing stage.
Acknowledgments
XAFS experiments were conducted at BL11S2 (Proposal No.202305004) and BL5S1 (Proposal No.202401006) of the Aichi Synchrotron Radiation Center, Aichi Science and Technology Foundation, Aichi, Japan.
- [1] World Steel Association, “Steel statistics yearbook 2023.” https://worldsteel.org/wp-content/uploads/Steel-Statistical-Yearbook-2023.pdf [Accessed June 21, 2025]
- [2] Y. Mizuno, Y. Kishita, S. Fukushige, and Y. Umeda, “Envisioning sustainable manufacturing industries of Japan,” Int. J. Automation Technol., Vol.8, No.5, pp. 634-643, 2014. https://doi.org/10.20965/ijat.2014.p0634
- [3] Nippon Slag Association (in Japanese). https://www.slg.jp/cms/wp-content/themes/original/pdf/report-2023.pdf [Accessed June 19, 2025]
- [4] Japan Cement Association (in Japanese). https://www.jcassoc.or.jp/cement/4pdf/jd6_01.pdf [Accessed June 21, 2025]
- [5] A. Aldrian, J. G. Raith, D. Hollen, and R. Pomberger, “Influence of chromium containing spinels in an electric arc furnace slag on the leaching behavior,” J. Solid Waste Technol. Manag., Vol.41, No.4, pp. 357-365, 2015. https://doi.org/10.5276/JSWTM.2015.357
- [6] Y. Ji, S. Shen, J. Liu, J. Guo, and Y. Zhao, “Mechanisms involved in the roasting of pellets composed of stainless steel slag and sodium hydroxide to extract chromium,” ISIJ Int., Vol.56, No.10, pp. 1751-1757, 2016. https://doi.org/10.2355/isijinternational.ISIJINT-2016-320
- [7] Z. Peng, L. Wang, F. Gu, H. Tang, M. Rao, Y. Zhang, G. Li, and T. Jiang, “Recovery of chromium from ferronickel slag: A comparison of microwave roasting and conventional roasting strategies,” Powder Technol., Vol.372, pp. 578-584, 2020. https://doi.org/10.1016/j.powtec.2020.05.103
- [8] C. Tokoro, S. Lim, Y. Sawamura, M. Kondo, K. Mochidzuki, T. Koita, T. Namihira, and Y. Kikuchi, “Copper/silver recovery from photovoltaic panel sheet by electrical dismantling method,” Int. J. Automation Technol., Vol.14, No.6, pp. 966-974, 2020. https://doi.org/10.20965/ijat.2020.p0966
- [9] C. Ye, Y. Takaya, Y. Tsunazawa, K. Mochidzuki, and C. Tokoro, “Influence of agitator shape on characteristics and grinding efficiency of attritor mill,” Int. J. Automation Technol., Vol.16, No.6, pp. 756-765, 2022. https://doi.org/10.20965/ijat.2022.p0756
- [10] K. Bru, A. Seron, A. Morillon, D. Algermissen, C. Lerouge, and N. Menad, “Characterization of a chromium-bearing carbon steel electric arc furnace slag after magnetic separation to determine the potential for iron and chromium recovery,” Minerals, Vol.12, No.1, Article No.47, 2022. https://doi.org/10.3390/min12010047
- [11] F. Kukurugya, P. Nielsen, and L. Horckmans, “Up-concentration of chromium in stainless steel slag and ferrochromium slags by magnetic and gravity separation,” Minerals, Vol.10, No.10, Article No.906, 2020. https://doi.org/10.3390/min10100906
- [12] L. Horckmans, R. Möckel, P. Nielsen, F. Kukurugya, C. Vanhoof, A. Morillon, and D. Algermissen, “Multi-analytical characterization of slags to determine the chromium concentration for a possible re-extraction,” Minerals, Vol.9, No.10, Article No.646, 2019. https://doi.org/10.3390/min9100646
- [13] Z. Yan, Q. Zhao, C. Han, X. Mei, C. Liu, and M. Jiang, “Effects of iron oxide on crystallization behavior and spatial distribution of spinel in stainless steel slag,” Int. J. Miner. Metall. Mater., Vol.31, pp. 292-300, 2024. https://doi.org/10.1007/s12613-023-2713-7
- [14] K. Izumi, Y. Takaya, and C. Tokoro, “Classification evaluation method considering the effect of specific gravity sorting in air classification – Case study of solar panel cell sheets classification –,” Kagaku Kogaku Ronbunshu, Vol.50, No.1, pp. 9-16, 2024 (in Japanese). https://doi.org/10.1252/kakoronbunshu.50.9
- [15] B. Ravela and M. Newvilleb, “ATHENA, ARTEMIS, HEPHAESTUS: Data analysis for X-ray absorption spectroscopy using IFEFFIT,” J. Synchrotron Radiat., Vol.12, No.4, pp. 537-541, 2005. https://doi.org/10.1107/S0909049505012719
- [16] S. Liang, “Numerical study of classification of ultrafine particles in a gas-solid field of elbow-jet classifier,” Chem. Eng. Commun., Vol.197, No.7, pp. 1016-1032, 2010. https://doi.org/10.1080/00986440903359392
- [17] J. W. French and W. G. Guntheroth, “An explanation of asymmetric upper extremity blood pressures in supravalvular aortic stenosis,” Circulation, Vol.42, No.1, pp. 31-36, 1970. https://doi.org/10.1161/01.cir.42.1.31
- [18] F. Engström, D. Adolfsson, Q. Yang, C. Samuelsson, and B. Björkman, “Crystallization behaviour of some steelmaking slags,” Steel Res. Int., Vol.81, No.5, pp. 362-371, 2010. https://doi.org/10.1002/srin.200900154
- [19] Q. Zeng, J. Li, Q. Mou, H. Zhu, and Z. Xue, “Effect of FeO on spinel crystallization and chromium stability in stainless steel-making slag,” JOM, Vol.71, No.7, pp. 2331-2337, 2019. https://doi.org/10.1007/s11837-019-03465-0
- [20] Z. Wang and I. Sohn, “Selective elemental concentration during the solidification of stainless steel slags for increased Cr recovery with MnO addition,” J. Ceram. Soc., Vol.103, No.10, pp. 6012-6024, 2020. https://doi.org/10.1111/jace.17296
- [21] J. Burja, F. Tehovnik, J. Medved, M. Godec, and M. Knap, “Chromite spinel formation in steelmaking slags,” Mater. Technol., Vol.48, No.5, pp. 753-756, 2014.
- [22] L. Cao, C. Liu, Q. Zhao, and M. Jiang, “Effect of Al2O3 modification on enrichment and chromium in stainless steel slag,” J. Iron Steel Res. Int., Vol.24, No.3, pp. 258-265, 2017. https://doi.org/10.1016/s1006-706x(17)30038-9
- [23] Q. Zhao, C. Liu, L. Cao, X. Zheng, and M. Jiang, “Effect of lime on stability of chromium in stainless steel slag,” Minerals, Vol.8, No.10, Article No.424, 2018. https://doi.org/10.3390/min8100424
- [24] L. Cao, C. Liu, Q. Zhao, and M. Jiang, “Growth behavior of spinel in stainless steel slag during cooling process,” J. Iron Steel Res. Int., Vol.25, No.11, pp. 1131-1139, 2018. https://doi.org/10.1007/s42243-018-0058-7
- [25] M. Tossavainen, F. Engstrom, Q. Yang, N. Menad, M. L. Larsson, and B. Bjorkman, “Characteristics of steel slag under different cooling conditions,” Waste Management, Vol.27, No.10, pp. 1335-1344, 2007. https://doi.org/10.1016/j.wasman.2006.08.002
- [26] S. Luo, X. Ma, W. Wang, L. Zhang, and Y. Sun, “Stabilities and transformations of manganese incorporated magnesium–chromium spinels,” Metall. Mater. Trans. B, Vol.55, pp. 4776-4786, 2024. https://doi.org/10.1007/s11663-024-03297-7
- [27] T. Kuwayama, A. Honda, M. Yamada, and T. Mise, “Utilization of hardening properties of reducing slag produced by electric furnace,” J. Jpn. Soc. Waste Manag., Vol.1, No.1, pp. 19-28, 1990 (in Japanese). https://doi.org/10.3985/jswme.1.19
- [28] M. A. Bredig, “Polymorphism of calcium orthosilicate,” J. Am. Ceram. Soc., Vol.33, No.6, pp. 188-192, 1950. https://doi.org/10.1111/j.1151-2916.1950.tb12789.x
- [29] Q. Zhao, C. Liu, L. Cao, X. Zheng, and M. Jiang, “Stability of chromium in stainless steel slag during cooling,” Minerals, Vol.8, No.10, Article No.445, 2018. https://doi.org/10.3390/min8100445
- [30] Y. Ji, S. Shen, J. Liu, S. Yan, Z. Zhang, and Y. Xue, “Extracting chromium from stainless steel slags by NaOH-added pellet roasting followed by water leaching,” Steel Res. Int., Vol.88, No.9, Article No.1600460, 2017. https://doi.org/10.1002/srin.201600460
 This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.
This article is published under a Creative Commons Attribution-NoDerivatives 4.0 Internationa License.